The Digital Fish Library
Featured Fish
By Kara Yopak, Ph.D.
Taxonomy
 A Sketch of the Great White from Compagno (1984).
All sharks are part of a large class of fishes called Chondrichthyes, or cartilaginous fishes, which is comprised of approximately 1100 extant species worldwide (Compagno, 1999). Cartilaginous fishes include chimaeras, skates, rays, and, of course, sharks. The great white shark, Carcharodon carcharias, is a member of the family Lamnidae, a group referred to as the mackerel sharks, comprised of other known species such as the shortfin mako shark (Isurus oxyrinchus) and the salmon shark (Lamna diptrosis). Members of Lamnidae are found globally, known for their large gill slits, fusiform bodies, and pronounced caudal keel, which enables them to be efficient, strong swimmers (Compagno, 1984).
A Sketch of the Great White from Compagno (1984).
All sharks are part of a large class of fishes called Chondrichthyes, or cartilaginous fishes, which is comprised of approximately 1100 extant species worldwide (Compagno, 1999). Cartilaginous fishes include chimaeras, skates, rays, and, of course, sharks. The great white shark, Carcharodon carcharias, is a member of the family Lamnidae, a group referred to as the mackerel sharks, comprised of other known species such as the shortfin mako shark (Isurus oxyrinchus) and the salmon shark (Lamna diptrosis). Members of Lamnidae are found globally, known for their large gill slits, fusiform bodies, and pronounced caudal keel, which enables them to be efficient, strong swimmers (Compagno, 1984).
Biology and Ecology
The great white shark is approximately 1.2 - 1.5m at birth and has been documented to grow up to lengths of 7.2 m (Compagno, 1998). They are ovoviviparous, giving birth to live young (Dulvy and Reynolds, 1997). These species are known to be oophagous as well, wherein the embryos feed on ova produced by the mother after the yolk sac is absorbed (Dulvy and Reynolds, 1997). These sharks are widely distributed around the world, but are abundantly found along the coast of California, South Africa, Australia, and New Zealand (Klimley et al., 1996; Compagno, 2001). The are primarily coastal animals, found close inshore at times, but have also been known to range down to the continental slope (Compagno, 1984). They are efficient cruisers, often swimming long distances, but have also been documented in breaching behavior, where they leap out of the water to feed on seals (Martin, 2007). They are highly versatile predators that feed on a wide range of prey, including teleost (or bony) fishes, squid, marine mammals, and other chondrichthyans [Compagno et al., 1989, Cortes, 1999].
Neuroanatomy of a Great White
Brain Organization
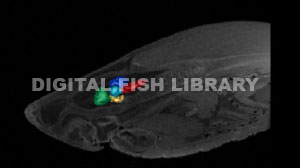 3D Segmentation of the great white brain acquired from MR images (Segmentation by Kara Yopak).
3D Segmentation of the great white brain acquired from MR images (Segmentation by Kara Yopak).
 Telencephalon - green, Diencephalon - yellow, Mesencephalon - cyan,
Cerebellum - blue,
Telencephalon - green, Diencephalon - yellow, Mesencephalon - cyan,
Cerebellum - blue,
Medulla - magenta.
In order to understand the behaviors of these animals, it’s critical to understand the neural mechanisms and structures moderating them. The five main structures of the shark brain include the telencephalon, diencephalon, mesencephalon, cerebellum, and medulla. It is critical to note that the structures of a chondrichthyan brain are mutimodal and not solely responsible for one sensory ability or behavior. The telencephalon (green), located in the forebrain, accounts for 39% of the brain of a great white (Yopak et al., 2007). This structure receives olfactory information and is also responsible for the integration of memory, learning, and spatial awareness and acts as a multisensory relay station of sorts, receiving input from the visual and octavolateralis (including electrosensory, auditory, and mechanosensory) systems (e.g. Ebbesson and Schroeder, 1971; Cohen et al., 1973; Graeber, 1978; Blackmann et al., 1987; Smeets and Northcutt, 1987). The telencephalon interprets sensory input from the diencephalon (yellow), which is engaged in controlling motivation, regulating the production of hormones, and relaying that information to the animal’s endocrine system (Northcutt, 1978; Hofmann, 1999) and occupies 6% of the great white shark brain (Yopak et al., 2007). The mesencephalon (cyan) receives input from the retina and houses the optic tectum (Hofmann, 1999). This structure is also implicated in integrating electrosensory and mechanosensory information (Bres, 1993; Tricas et al., 1997) and may be responsible for the behavioral responses to novel visual stimuli (Bodznick, 1991). Based on a data set of 51 different shark species, great whites demonstrate a relatively large mesencephalon, accounting for approximately 14% of the brain (Yopak et al., 2007). The medulla (magenta) contains the cerebellar-like structures, which receive electrosensory, lateral line, acoustic, and vestibular input (Hofmann, 1999). The medulla is also involved in a shark’s homeostasis and acts as a relay station between the brain and spinal cord (Northcutt, 1978; Smeets et al., 1983; Kotrschal et al., 1998; Hofmann, 1999). This structure occupies 24% of the brain of a great white shark, which is slightly large compared to other species.
The Cerebellum
_300_wp.jpg) An MRI cross section of the great white cerebellum.One area of particular interest is the brain structure known as the cerebellum (blue), which accounts for 18% of the brain of C. carcharias, and whose function is highly controversial. This structure appeared first in early chondrichthyans (Butler, 2003) and exhibits substantial variation in both the degree of folding and symmetry (Northcutt, 1977, 1978; Yopak et al., 2007). A large body of research suggests that it modulates motor tasks (Paul and Roberts, 1979; New, 2001) and error correction (Montgomery et al., 2002). However, other evidence sows that the cerebellum may be involved in coordination of target tracking and analysis of the consequence of an organism’s own movement, rather than control of these movements themselves (Paulin, 1993, 1997). Current research is investigating comparative brain morphology in a wide range of species, and linking these variations to dimensionality of the environment and/or agile prey capture (Yopak et al., 2007). The great white, in particular, has a highly foliated cerebellum, which may be linked to the 3-dimensional oceanic environment in which it lives and its predation on highly active, agile prey.
An MRI cross section of the great white cerebellum.One area of particular interest is the brain structure known as the cerebellum (blue), which accounts for 18% of the brain of C. carcharias, and whose function is highly controversial. This structure appeared first in early chondrichthyans (Butler, 2003) and exhibits substantial variation in both the degree of folding and symmetry (Northcutt, 1977, 1978; Yopak et al., 2007). A large body of research suggests that it modulates motor tasks (Paul and Roberts, 1979; New, 2001) and error correction (Montgomery et al., 2002). However, other evidence sows that the cerebellum may be involved in coordination of target tracking and analysis of the consequence of an organism’s own movement, rather than control of these movements themselves (Paulin, 1993, 1997). Current research is investigating comparative brain morphology in a wide range of species, and linking these variations to dimensionality of the environment and/or agile prey capture (Yopak et al., 2007). The great white, in particular, has a highly foliated cerebellum, which may be linked to the 3-dimensional oceanic environment in which it lives and its predation on highly active, agile prey.
Great White Conservation
 This juvenile great white was caught, tagged, and released as part of an ongoing tracking study.
Famous in reputation but not often in fact, the great white shark is known for being a voracious Hollywood monster. Attacks on humans are believed by experts to be a case of mistaken identity, where the human is mistook for the shark’s natural prey (FAO, 1988), though these attacks are often sensationalized in the media. Great whites are unique in their remarkable predatory skills and highly evolved sensory systems, yet it is their apex position in most marine food webs that makes them both formidable and ecologically necessary. Due to their notoriety, the jaws, fins, and teeth of these sharks are collected and illicitly traded and as a result, coupled with its late sexual maturity and small litter size (Gilmore, 1993, Smith et al., 1998), these ecologically critical species are listed as Vulnerable on the IUCN Red List of Threatened Species (www.IUCNredlist.org). Originally thought to be solely coastal species associated with continental and insular shelves (Compagno, 1984), recent studies on great white spatial dynamics have shown transoceanic return migrations as well (Bonfil et al., 2005). Studies such as these stress the importance of reassessing management and conservation strategies of these threatened species, as great whites are now documented to leave domestically protected coastal waters into the open ocean, increasing their mortality risks (Bonfil et al., 2005).
This juvenile great white was caught, tagged, and released as part of an ongoing tracking study.
Famous in reputation but not often in fact, the great white shark is known for being a voracious Hollywood monster. Attacks on humans are believed by experts to be a case of mistaken identity, where the human is mistook for the shark’s natural prey (FAO, 1988), though these attacks are often sensationalized in the media. Great whites are unique in their remarkable predatory skills and highly evolved sensory systems, yet it is their apex position in most marine food webs that makes them both formidable and ecologically necessary. Due to their notoriety, the jaws, fins, and teeth of these sharks are collected and illicitly traded and as a result, coupled with its late sexual maturity and small litter size (Gilmore, 1993, Smith et al., 1998), these ecologically critical species are listed as Vulnerable on the IUCN Red List of Threatened Species (www.IUCNredlist.org). Originally thought to be solely coastal species associated with continental and insular shelves (Compagno, 1984), recent studies on great white spatial dynamics have shown transoceanic return migrations as well (Bonfil et al., 2005). Studies such as these stress the importance of reassessing management and conservation strategies of these threatened species, as great whites are now documented to leave domestically protected coastal waters into the open ocean, increasing their mortality risks (Bonfil et al., 2005).
Literature Cited
Bleckmann, H., T.H. Bullock, and J.M. Jørgensen. (1987) The lateral line mechanoreceptive mesencephalic, diencephalic, and telencephalic regions in the thornback ray, Platyrhinoidis triseriata (Elasmobranchii). Journal of Comparative Physiology A. 161: p. 459-474.
Bodznick, D. (1991) Elasmobranch vision: Multimodal integration in the brain. Journal of Experimental Zoology Supplement. 256(S5): p. 10
Bonfil, R., M. Meÿer, M.C. Scholl, R. Johnson, S. O'Brien, H. Oosthuizen, S. Swanson, D. Kotze, and M. Paterson. (2005) Transoceanic Migration, Spatial Dynamics, and Population Linkages of White Sharks. Science. 310(5745): p. 100-103.
Bres, M. (1993) The behaviour of sharks. Reviews in Fish Biology and Fisheries. 3(2): p. 133-159.
Butler, A.B. (2003) Sensory systems and brain evolution across the bilateria: Commonalities and constraints, In Sensory Processing in Aquatic Environments, S.P. Collin and N.J. Marshall, Editors. Springer-Verlag: New York, NY. p. 375-388.
Cohen, J.L., T.A. Duff, and S.O.E. Ebbesson. (1973) Electrophysiological identification of a visual area in the shark telencephalon. Science. 182: p. 492-494.
Compagno, L.J.V. (1984) FAO Species Catalogue. Sharks of the world. An annotated and illustrated catalogue of shark species known to date. I. Hexanchiformes to Lamniformes. Vol. 4. Rome, Italy: FAO Fisheries Synopsis. 249.
Compagno, L.J.V. (1998) Lamnidae. Mackerel sharks, makos, white sharks, porbeagles, In FAO Identification Guide for Fishery Purposes. The Living Marine Resources of the Western Central Pacific, K.E. Carpenter and V.H. Niem, Editors. FAO Fisheries Synopsis: Rome, Italy. p. 1274-1278.
Compagno, L.J.V. (1999) Checklist of living elasmobranches, In Sharks, Skates, and Rays: The Biology of Elasmobranch Fishes, W.C. Hamlet, Editor. Johns Hopkins University Press: Baltimore, MD. p. 471-498.
Compagno, L.J.V. (2001) FAO Species Catalogue. Sharks of the world. And annotated and illustrated catalogue of shark species known to date. Bullhead, mackerel, and carpet sharks (Heterodontiformes, Lamniformes and Orectolobiformes). Vol. 2. Rome, Italy: FAO Fisheries Synopsis.
Compagno, L.J.V., D.A. Ebert and M.J. Smale, 1989. Guide to the sharks and rays of southern Africa. New Holland (Publ.) Ltd., London. 158 p.
Cortés, E. (1999) Standardized diet compositions and trophic levels of sharks. ICES Journal of Marine Science. 56(5): p. 707-717.
Dulvy, N.K. and J.D. Reynolds. (1997) Evolutionary transitions among egg-laying, live-bearing, and maternal inputs in sharks and rays. Proceedings of the Royal Society, London. B. 264: p. 1309-1315.
Ebbesson, S.O.E. and D. Schroeder, M. (1971) Connections of the nurse shark's telencephalon. Science. 173(3993): p. 254-256.
FAO (1988) Yearbook of fishery statistics 1986: Catches and landings. FAO Fish.eries Series. 62: 479 p.
Gilmore, R.G. (1993) Reproductive biology of lamnoid sharks. Environmental Biology of Fishes. 38(1/3): 95-114.
Graeber, R.C. (1978) Behavioral studies correlated with central nervous system integration of vision in sharks, In Sensory Biology of Sharks, Skates, and Rays, E.S. Hodgson and R.F. Mathewson, Editors. U.S. Government Printing Office: Washington DC. p. 195-225.
Hofmann, M.H. (1999) Nervous system, In Sharks, Skates, and Rays: The Biology of Elasmobranch Fishes, W.C. Hamlet, Editor. Johns Hopkins University Press: Baltimore, MD. p. 273-299.
Kotrschal, K., M.J. van Staaden, and R. Huber. (1998) Fish brains: Evolution and environmental relationships. Reviews in Fish Biology and
Linnaeus, C. (1758) Systema Naturae, Ed. X. (Systema naturae per regna tria naturae, secundum classes, ordines, genera, species, cum characteribus, differentiis, synonymis, locis. Tomus I. Editio decima, reformata.) Holmiae. Systema Nat. ed. 10 i-ii + 1-824
Martin, R.A. (2007) A review of shark agonistic displays: Comparison of display features and implications for shark-human interactions. Marine and Freshwater Behaviour and Physiology. 40(1): p. 3-34.
Montgomery, J.C., G. Carton, and D. Bodznick. (2002) Error-driven motor learning in fish. Biological Bulletin. 203: p. 238-239.
New, J.G. (2001) Comparative neurobiology of the elasmobranch cerebellum: Theme and variations on a sensorimotor interface. Environmental Biology of Fishes. 60: p. 93-108.
Northcutt, R.G. (1977) Elasmobranch central nervous system organization and its possible evolutionary significance. American Zoologist. 17: p. 411-429.[1]
Northcutt, R.G. (1978) Brain organization in the cartilaginous fishes, In Sensory Biology of Sharks, Skates, and Rays, E.S. Hodgson and R.F. Mathewson, Editors. Office of Naval Research: Arlington, VA. p. 117-194.
Paul, D.H. and B.L. Roberts. (1979) The significance of cerebellar function for a reflex movement of the dogfish. Journal of Comparative Ph
Paulin, M.G. (1993) The role of the cerebellum in motor control and perception. Brain, Behavior, and Evolution. 41(1): p. 39-51.
Paulin, M.G. (1997) Neural representations of moving systems. International Review of Neurobiology. 41: p. 515-533.
Smeets, W.J.A.J., R. Nieuwenhuys, and B.L. Roberts. (1983) The Central Nervous System of Cartilaginous Fishes: Structural and Functional Correlations. New York, NY: Springer-Verlag. 266.
Smeets, W.J.A.J. and R.G. Northcutt. (1987) At least one thalamotelencephalic pathway in cartilaginous fishes projects to the medium pallium. Neuroscience Letters. 78: p. 277-282.
Smith, S.W., Au, D.W., Show, C. (1998) Intrinsic rebound potential of 26 species of Pacific sharks. Marine and Freshwater Research. 49:663-678
Tricas, T.C., K. Deacon, P. Last, J.E. McCosker, T.I. Walker, and L. Taylor. (1997) Sharks and Rays, ed. L. Taylor. Sydney, Australia: Reader's Digest.
Yopak, K.E., T.J. Lisney, S.P. Collin, and J.C. Montgomery. (2007) Variation in brain organization and cerebellar foliation in chondrichthyans: Sharks and holocephalans. Brain, Behavior, and Evolution. 69(4): p. 280-300.

